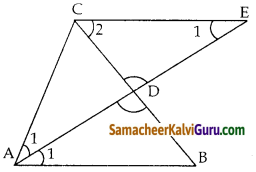
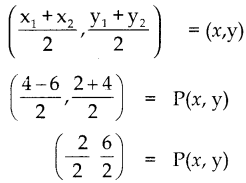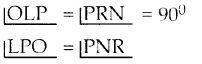Tamilnadu State Board New Syllabus Samacheer Kalvi 10th Maths Guide Pdf Chapter 5 ஆயத்தொலை வடிவியல் Ex 5.1 Textbook Questions and Answers, Notes.
TN Board 10th Maths Solutions Chapter 5 ஆயத்தொலை வடிவியல் Ex 5.1
கேள்வி 1.
கீழ்க்கண்ட புள்ளிகளால் அமைக்கப்படும் முக்கோணத்தின் பரப்பு காண்க. i) (1, -1), (-4, 6) மற்றும் (-3, -5)
தீர்வு :
படத்தில் கொடுக்கப்பட்ட புள்ளிகளைக் கடிகார முள்ளின் எதிர் திசையில் அமையுமாறு குறிக்க வும். A (1, – 1), B (-4, 6), C (-3,-5) என்பன முக்கோணத்தின் முனைகள் என்க.
ΔABCன் பரப்பு
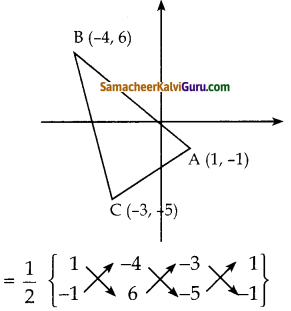
= \(\frac { 1 }{ 2 }\) {(6+20+3) – (4-18-5)}
= \(\frac { 1 }{ 2 }\)(29- (-19) = \(\frac { 1 }{ 2 }\) [29 + 19)
= \(\frac { 1 }{ 2 }\) x 48
= 24 ச.அலகுகள்
![]()
ii) (-10, -4), (-8, -1) மற்றும் (-3, -5)
தீர்வு :
படத்தில் கொடுக்கப்பட்ட புள்ளிகளைக் கடிகார முள்ளின் எதிர் திசையில் அமையுமாறு குறிக்கவும்.
A(-10, -4), B(-8, -1) என்ப ன C(-3, -5)
முக்கோணத்தின் முனைகள் என்க.
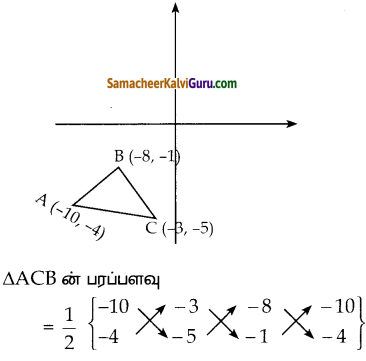
ΔACB ன் பரப்பளவு
= \(\frac { 1 }{ 2 }\) {(50+3+32) – (12+40+10)}
= \(\frac { 1 }{ 2 }\)[85 – 62]
= \(\frac { 1 }{ 2 }\) x 23
= 11.5 ச. அலகுகள்
கேள்வி 2.
கீழ்க்காணும் புள்ளிகள் ஒரே நேர்க்கோட்டில் அமையுமா என தீர்மானிக்கவும்.
i) (-\(\frac { 1 }{ 2 }\), 3), (-5, 6) மற்றும் (-8, 8)
தீர்வு :
A(-\(\frac { 1 }{ 2 }\),3), B(-5, 6), C(-8,8) ஆகியன
கொடுக்கப்பட்ட புள்ளிகள் ஆகும்.
ΔABC ன் பரப்பு
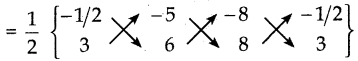
= \(\frac { 1 }{ 2 }\) {(-3-40-24) – (-15-48-4)}
= \(\frac { 1 }{ 2 }\)[-67 – (-67)]
= \(\frac { 1 }{ 2 }\)[-67 + 67)] = 0
எனவே, கொடுக்கப்பட்ட புள்ளிகள் ஒரே நேர்க்கோட்டில் அமைந்துள்ளன.
ii) (a,b+c), (b, c+a) மற்றும் (c, a+b)
தீர்வு :
A(a, b+c), B(b, c+a), C(c, a+b) ஆகியன கொடுக்கப்பட்ட புள்ளிகள் ஆகும்.
ΔABC ன் பரப்பு
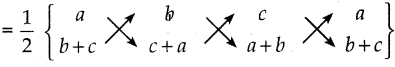
= \(\frac { 1 }{ 2 }\) {{a(c+a) + b(a+b) + c[b+c)) – (b(b+c) + c(c+a) = a(a+b))}
= \(\frac { 1 }{ 2 }\) [ac + a2 + ba + b + cb + c2 – [b2 + bc + c2 + ac + a2 + ab)]
= \(\frac { 1 }{ 2 }\) [ac + a2 + ba + b + cb + c2 – b2 – bc – c2 – ac – a2 – ab]
= \(\frac { 1 }{ 2 }\) x 0 = 0
எனவே, கொடுக்கப்பட்ட புள்ளிகள் ஒரே நேர்க்கோட்டில் அமைந்துள்ளன.
![]()
கேள்வி 3.
வரிசையில் அமைந்த முக்கோணத்தின் முனைப்புள்ளிகளும், அதன் பரப்புகளும் அட்டவணையில் கொடுக்கப்பட்டுள்ளன.’p’ யின் மதிப்பைக் காண்க.

தீர்வு :
i) A(0,0), B(p,8), C(6,2) ஆகியன கொடுக்கப்பட்ட முக்கோணத்தின் முனைப்புள்ளிகள் ஆகும்.
ΔABC ன் பரப்பளவு = 20 ச.அ
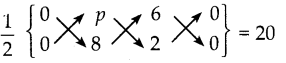
[0 + 2p + 0] – [0 + 48 + 0] = 20 x 2
2p – 48 = 40
2p = 40 + 48
P = \(\frac{88}{2}\)
p = 44
ii) A(p, p), B(5, 6), C(5, -2) என்பன முக்கோணத்தின் முனைப்புள்ளிகள் ஆகும்.
ΔABC ன் பரப்பு = 32 ச.அ
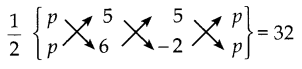
[6p – 10 + 5p] – [5p + 30 – 2p] = 32 x 2
(11p – 10) – (3p + 30) = 32 x 2
11p- 10 – 3p – 30 = 32 x 2
8p-40 = 32 x 2
8p = 64 + 40
8p = 104
p = 104/8
p = 13
கேள்வி 4.
கொடுக்கப்பட்ட புள்ளிகள் ஒரே நேர்க்கோட்டில் அமைந்தவை எனில், ‘a’ யின் மதிப்பைக் காண்க.
i) (2, 3), (4, a) மற்றும் (6,-3)
ii) (a,2-2a),(-a+1, 2a) மற்றும் (-4-a,6-2a)
தீர்வு :
A (2, 3), B (4, a) மற்றும் (6, -3) ஆகிய கொடுக்கப்பட்ட புள்ளிகள் ஒரே நேர்க்கோட்டில் அமையும்.
எனவே, ΔABC ன் பரப்பளவு = 0
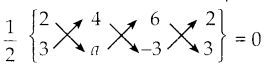
(2a-12+18) – (12-6a-6) = 0 x 2 = 0
(2a+6) – (6 – 6a) = 0
2a + 6 – 6 + 6a = 0
8a = 0
a = 0
ii) A (a, 2-2a), B (-a+1, 2a) மற்றும் C (-4-2, 6-2a) ஆகிய கொடுக்கப்பட்ட புள்ளிகள் ஒரே நேர்க்கோட்டில் அமையும். எனவே, ΔABC ன் பரப்பளவு = 0
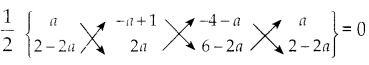
\(\frac { 1 }{ 2 }\) {(2a-+(-a+1)(6-2a)+(-4-a) (2-2a))
((2-2a)(-a+1)+(2a)(-4-a)+a(6-2a})} = 0
(2a2 – 6a + 2a2 + 6 – 2a – 8 + 8a – 2a + 2a2 )-(-2a + 2 + 2a2 – 2a – 8a – 2a2 + 6a – 2a2)} = 0 x 2 = 0
(6a2 – 2a – 2) – (-2a2 – 6a + 2) = 0
6a2 – 2a – 2 + 2a2 + 6a – 2 = 0
8a2 + 4a – 4 = 0
2a2 + a – 1 = 0
(2a-1) (a+1) = 0
a = \(\frac { 1 }{ 2 }\), -1
a என் மதிப்பு \(\frac { 1 }{ 2 }\) (அல்லது) -1
![]()
கேள்வி 5.
கொடுக்கப்பட்ட புள்ளிகளை முனைகளாக கொண்ட நாற்கரத்தின் பரப்பைக் காண்க?
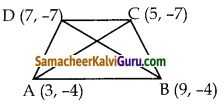
i) (-9, -2), (-8, -4) (2, 2) மற்றும் (1, – 3)
ii) (-9, 0), (-8, 6) (-1, -2) மற்றும் (-6,-3)
தீர்வு :
i) நாற்கரத்தின் பரப்பைக் காண்பதற்கு முன்பாகக் கொடுக்கப்பட்ட புள்ளிகளை வரைபடத்தில் குறிக்க வேண்டும். A(-9, -2), B(-8, -4), C(2, 2) மற்றும் D(1, -3) என்பன முனைப்புள்ளிகள் ஆகும்.
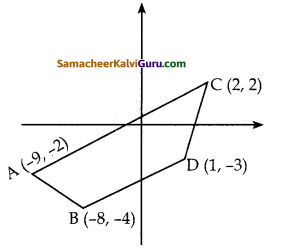
எனவே, நாற்கரம் ABDC ன் பரப்பு
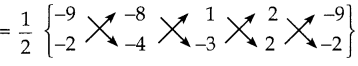
= \(\frac { 1 }{ 2 }\) {(36+24+2-4) – (16-4-6-18)}
= \(\frac { 1 }{ 2 }\) {58 – (-12)}
= \(\frac { 1 }{ 2 }\) (58 +12) = \(\frac { 1 }{ 2 }\) x 70
= 35 ச. அலகுகள்
நாற்கரத்தின் பரப்பளவு = 35 ச. அலகுகள்
ii) நாற்கரத்தின் பரப்பைக் காண்பதற்கு முன்பாகக் கொடுக்கப்பட்ட புள்ளிகளை வரைபடத்தில் குறிக்க வேண்டும். புள்ளிகளை எதிர் கடிகார திசையில் எடுத்து கொள்க. A (-9, 0), B (-8, 6), C (-1, – 2) மற்றும் D (-6, -3 என்பன முனைப்புள்ளிகள் ஆகும்.
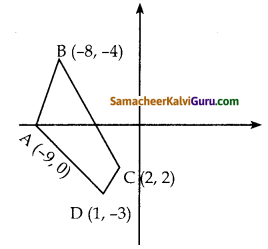
எனவே நாற்கரத்தின் பரப்பளவு (ADCB)
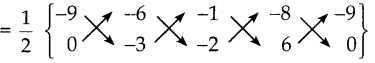
= \(\frac { 1 }{ 2 }\){(27+12-6+0) – (0+3+16-54)}
= \(\frac { 1 }{ 2 }\) [33 – (-35) = \(\frac { 1 }{ 2 }\)(33 + 35)
= \(\frac { 1 }{ 2 }\) x 68
= 34 ச.அலகுகள்
நாற்கரத்தின் பரப்பளவு = 34 ச. அலகுகள்
![]()
கேள்வி 6.
(-4, -2), (-3, k), (3, -2) மற்றும் (2, 3) ஆகியவற்றை முனைகளாகக் கொண்ட நாற்கரத்தின் பரப்பு 28 சதுர அலகுகள் எனில், K ன் மதிப்பைக் காண்க.
தீர்வு :
நாற்கரத்தின் முனைகள் (-4, – 2), (-3, k), (3, -2) மற்றும் (2, 3). மற்றும் அதன் பரப்பு 28 ச.அலகுகள்
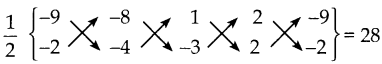
\(\frac { 1 }{ 2 }\)(-4k+6+9-4) – (6+3k-4-12)} = 28
(-4k+11) – (3k – 10) = 28 x 2 -4k+11-3k+10 = 56
-7k + 21 = 56
-7k = 56-21
k = \(\frac{35}{-7}\)
k = -5
கேள்வி 7.
A(-3, 9), B (a, b) மற்றும் C(4, – 5) என்ப ன ஒரு கோட்டமைந்த புள்ளிகள் மற்றும் a+b = 1, எனில், a மற்றும் b யின் மதிப்பைக் காண்க.
தீர்வு :
கொடுக்கப்பட்டுள்ள மூன்று புள்ளிகள் ஒரே கோட்டமைந்த புள்ளிகள் ஆகும். எனவே. பரப்பளவு = 0
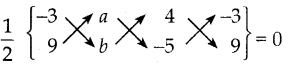
(-3b-5a+36) – (9a+4b+15) = 0 x 2
-3b – 5a + 36 – 9a – 4b – 15 = 0
-14a-7b+21 = 0
÷by (-7) 2a+b-3 = ) —–(1)
கொடுக்கப்பட்டது a+b = 1 ——-(2)
(1) – (2)
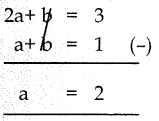
a = 2 ஐ சமன்பாடு (2) ல் பிரதியிட
a + b = 1
2 + b = 1
b = 1 – 2 = -1
b = -1
விடை : a = 2, b = -1
கேள்வி 8.
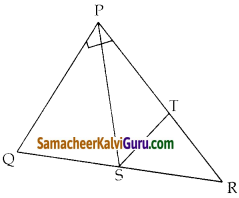
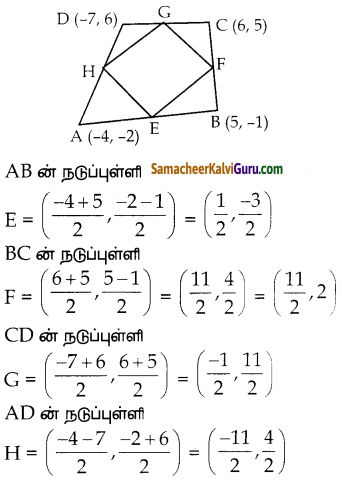
ΔABC ன் பக்கங்கள் AB, BC மற்றும் AC ஆகியவற்றின் நடுப்புள்ளிகள் முறையே P(11,7) , Q(13.5, 4) மற்றும் R(9.5, 4) என்க . முக்கோணத்தின் முனை புள்ளிகள் A,B மற்றும் C காண்க. மேலும், AABC – யின் பரப்பை APQR யின் பரப்புடன் ஒப்பிடுக.
தீர்வு :
முக்கோணத்தின் முனைகளை A (x1, y1), B (x2, y2) மற்றும் C(x3, y3) என்க.
P,Q, R என்பன AB, BC, CA என்ற முக்கோண பக்கங்களின் நடுப்புள்ளிகள் ஆகும்.
AB ன் நடுப்புள்ளி = P
\(\left(\frac{x_{1}+x_{2}}{2}, \frac{y_{1}+y_{2}}{2}\right)\) = (11, 7)
x மற்றும் y ஐ சமன்படுத்துக.
\(\frac{x_{1}+x_{2}}{2}\) = 11; \(\frac{y_{1}+y_{2}}{2}\) = 7
x1 + x2 = 22 —-(1); y1 + y2 = 14 —–(2)
இதே போல்
x2 + x3 = 19 —-(3);
y2 + y3 = 8 —-(4)
x3 + x1 = 27 —-(5);
y3 + y1 = 8 —–(6)
சேர்த்து (2)+(4)+(6)
= 2y1 + 2y2 + 2y3 = 30
= y1 + y2 + y3 = 15 ——(8)
சமன்பாடு (3) ஐ சமன்பாடு (7)ல் பிரதியிட
x1 + x2 + x3 = 34
x1 + 19 = 34
x1 = 34 – 19
x1 = 15
x1 = 15 ஐ சமன்பாடு (5)ல் பிரதியிட
x3 + x1 = 27
x3 + 15 = 27
x3 = 27 – 15
x3 = 12
x3 = 12 ஐ சமன்பாடு (3) ல் பிரதியிட,
x2 + x3 = 19
x2 + 12 = 19
x2 = 19 – 12)
சமன்பாடு (4) ஐ சமன்பாடு (8) ல் பிரதியிட
y1 + y2 + y3 = 15
y1 + 8 = 15
y1 = 15 – 8
y1 = 7
y1 = 7 ஐ சமன்பாடு (6)ல் பிரதியிட
y3 + y1 = 8
y3 + 7 = 8
y3 = 8 – 7
y3 = 1
y3 = 1 சமன்பாடு (4) ல் பிரதியிட
y2 + y3 = 8
y2 + 1 = 8
y2 = 8 – 1
y2 = 7
முக்கோணத்தின் முனைகள் A(15, 7), B(7, 7), மற்றும் C(12, 1)
ΔABC ன் பரப்பு
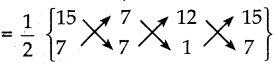
= \(\frac { 1 }{ 2 }\) {(105+7+84) – (49+84+15)}
= \(\frac { 1 }{ 2 }\) (196 – 148) = \(\frac { 1 }{ 2 }\) x 48 = 24 அலகுகள்
Δ POR ன் பரப்பு
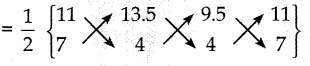
= \(\frac { 1 }{ 2 }\) {(44+54+66.5) – (94.5+38+44)}
= \(\frac { 1 }{ 2 }\) (164.5 – 176.5)
= \(\frac { 1 }{ 2 }\) x (-12) = -6
(பரப்பளவு ஒரு போதும் குறை எண்ணாக இருக்க இயலாது. எனவே, இதன் பரப்பை மிகை எண்ணாக எடுத்துக்கொள்ள வேண்டும்.)
ΔABC ன் பரப்பு = 4 x ΔPQR ன் பரப்பு
24 = 4 x 6
24 = 24 நிரூபிக்கப்பட்டது
![]()
கேள்வி 9.
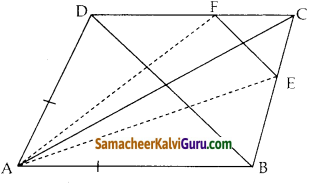
நாற்கர வடிவ நீச்சல் குளத்தின் கான்கிரீட் உள்முற்றமானது படத்தில் காட்டியுள்ள படி அமைக்கப்பட்டுள்ளது எனில், உள்முற்றத்தின் பரப்பு காண்க.

தீர்வு :
கொடுக்கப்பட்ட முனைகள் A(-4, -8), B(8, -4), C(6, 10) மற்றும் D(-10, 6)ன் பரப்பு
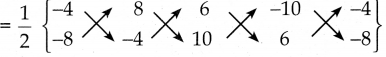
= \(\frac { 1 }{ 2 }\) {(16+80+36+80) – (-64-24-100-24)}
= \(\frac { 1 }{ 2 }\) 212 x 424
= 212 ச. அலகுகள்
கொடுக்கப்பட்ட முனைகள் E(-3, -5), F(6, -2), G(3, 7) மற்றும் H(-6, 4) ன் பரப்பு
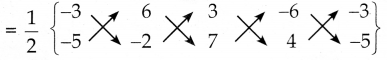
= \(\frac { 1 }{ 2 }\) {(6 + 42 + 12 + 30) – (-30 – 6 – 42 – 12)}
= \(\frac { 1 }{ 2 }\) x 180
= 90 ச.அலகுகள்
உளமுற்றத்தின் பரப்பு = ABCD ன் பரப்பு – EFGH ன் பரப்பு
= 212 – 90 = 122 ச.அலகுகள்
கேள்வி 10.
A(-5, -4) , B(1, 6) மற்றும் C(7, -4) ஆகியவற்றை முனைப்புள்ளிகளாகக் கொண்ட முக்கோணவடிவக் கண்ணாடிக்கு வர்ணம் பூசப்படுகிறது. 6 சதுர அடி பரப்புக்கு வர்ணம் பூச ஒரு வாளி தேவைப்படுகிறது எனில் கண்ணாடியின் முழுப்பகுதியையும் ஒரு முறை வர்ணம் பூச எத்தனை வாளிகள் தேவைப்படும்?
தீர்வு : கொடுக்கப்பட்ட முனைப்புள்ளிகளை வரைபடத்தில் குறித்து எதிர் கடிகார திசையில் எடுத்து கொள்க.
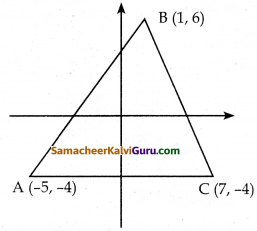
ΔACB ன் பரப்பு
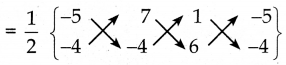
= \(\frac { 1 }{ 2 }\){(20+42-4) – (-28-4-30)}
= \(\frac { 1 }{ 2 }\)[58 – (-62]] = \(\frac { 1 }{ 2 }\) (58 + 62)
= \(\frac { 1 }{ 2 }\) x 120
= 60 ச.அலகுகள்
6 ச.அடி பரப்புக்கு வர்ணம் பூச ஒரு வாளி தேவைப்படுகிறது.
கண்ணாடி முழுவதும் வர்ணம் பூச தேவைப்படும் வாளிகள் = 60/6 = 10 வாளிகள்
10 வாளிகள் தேவைப்படும்.
![]()
கேள்வி 11.
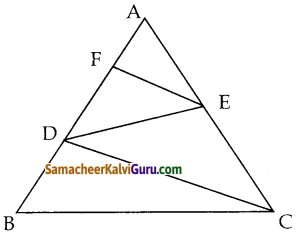
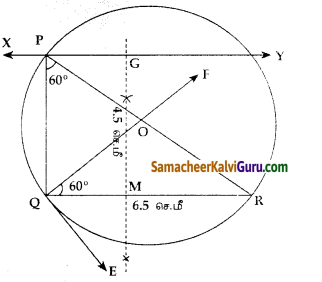
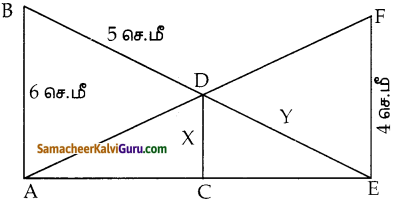
படத்தை பயன்படுத்திப் பரப்பைக் காண்க.
(i) முக்கோணம் AGF
(ii) முக்கோணம் FED
(iii) நாற்கரம் BCEG.
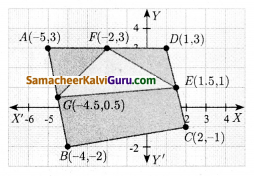
தீர்வு :
i) கொடுக்கப்பட்ட A(-5, 3), G(-4.5, 0.5) மற்றும் F(-2, 3) ஆகியவை முக்கோணத்தின் முனைகள்.
ΔAGFன் பரப்பளவு
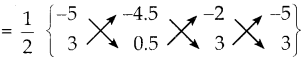
= \(\frac { 1 }{ 2 }\) {(-2.5-13.5-6) – (-13.5-1-15)}
= \(\frac { 1 }{ 2 }\) {–22 – (-29.5)}
= \(\frac { 1 }{ 2 }\) x 75
= 3.75 ச.அலகுகள்
ii) முக்கோணம் FEDன் பரப்பு முனைகள் F(-2, 3), E(1.5, 1) மற்றும் D(1, 3)
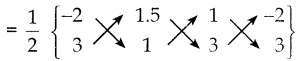
= \(\frac { 1 }{ 2 }\){(-2+4.5+3) – (-4.5+1-6)}
= 3 ச. அலகுகள்
iii) நாற்கரத்தின் முனைகள் B(-4,-2), C(2,-1), E(1.5, 1) மற்றும் G(-4.5, 0.5)
நாற்கரம் BCEG ன் பரப்பு
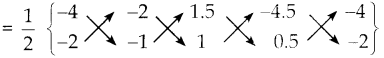
= \(\frac { 1 }{ 2 }\) {(4+2+0.75+9) – (-4-1.5-4.5-2}}
= \(\frac { 1 }{ 2 }\) {15.75 – (-12)}
= \(\frac { 1 }{ 2 }\) x {15.75+12} = \(\frac { 1 }{ 2 }\) x 27.75
= 13.875 ச. அலகுகள்