Tamilnadu Samacheer Kalvi 11th Chemistry Notes Chapter 4 Hydrogen Notes
Occurrence of Hydrogen – the sun and stars are composed of 85—95% Hydrogen. It is present in the form of water, organic compounds and in all lIving matter.
Occurrence of Hydrogen – the sun and stars are composed of 85—95% Hydrogen. It is present in the form of water, organic compounds and in all lIving matter.
H → f electrically neutral
H+ → Proton
H– → Hydride
H2 → diatomic gaseous molecule.
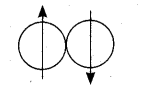
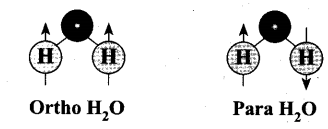
Ortho hydrogen – The hydrogen molecule in which the nuclear spins of the two hydrogen atoms are in the same side. Magnetic moment of it is twice.
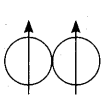
Para hydrogen – The hydrogen molecule in which the nuclear spins of the two hydrogen atoms are in the opposite side. Magnetic moment of it is zero.
Isotopes of hydrogen – Protium \({ }_{1}^{1} \mathrm{H}\), Deuterium ,\({ }_{1}^{2} \mathrm{H}\) and Tritium \({ }_{1}^{3} \mathrm{H}\).
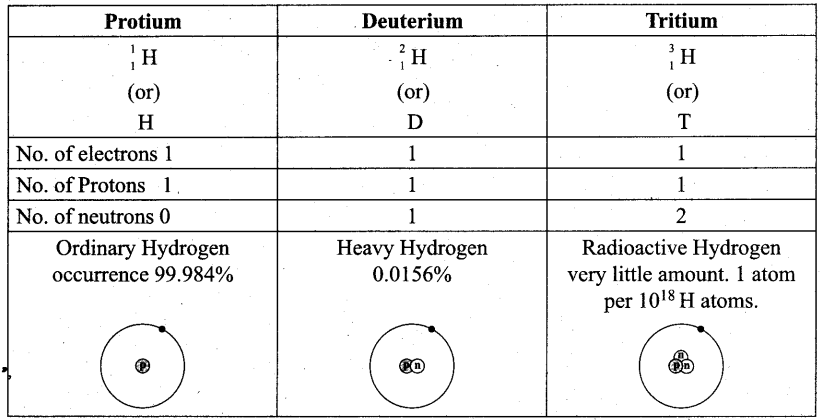
Half life period of Tritium – 12.33 years.
Isotopic effect – In a chemical reaction, when one of the atoms in the reactants is replaced by one of its isotopes.
Uses of Deuterium – It is used to study the movement of ground water and the hydrogen atom transfer mechanism in chemical reactions.
Uses of Tritium – It is used as a tracer element and it find applications such as emergency exit signs, illumination of wrist watches.
Preparation of Tritium – It occurs naturally as a result of nuclear reactions induced by cosmic rays in the upper atmosphere.
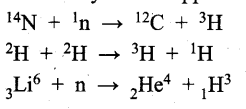
Preparation of Hydrogen
- Electrolysis of water.
- Action of dilute acids with metals like zinc, iron, tin.
- Steam reforming of hydrocarbons.
- Reaction of steam with red hot coke.
- Reaction of carbon monoxide with water.
- Cracking of long chain hydrocarbons.
Properties of hydrogen – It is a colourless, odourless, tasteless, lightest and inflammable gas.
Uses of hydrogen
- In the manufacture of Ammonia and Nitric acid.
- In the synthesis of methanol.
- As hydrogenating agent.
- As reducing agent in metallurgy.
- Calcium hydride is used as desiccant (drying agent).
- NaBH4, LiAlH4 used as reducing agent in organic chemistry.
- Atomic hydrogen and oxyhydrogen torches for cutting and welding.
- Liquid hydrogen is used as rocket fuel.
- H2 is used in fuel cells for generating electricity.
Water – H2O. It is the universal solvent. It occurs in the form of rivers and oceans (97.33%).
Ortho H2O and Para H2O – At the temperature conditions of the earth (300K), the OPR of H2O is 3. It is known that the OPR of water in interstellar clouds and comets has more para- H2O (OPR = 2.5) than on Earth.
Physical properties – Water is a colourless and volatile liquid. Hydrogen bonding responsible for high melting and boiling points of water.
Chemical properties – Water reacts with metals, non-metals and compounds.
Hard water – Water containing high amounts of bicarbonate, chloride and sulphate of magnesium and calcium is called hard water.
Soft water – Water containing soluble salts of calcium and magnesium is called soft water.
Soap – Sodium or Potassium salts of long chain fatty acids.
Temporary hardness – It is due to the presence of soluble bicarbonates of magnesium and calcium. It is removed by heating or by treating with lime.
Permanent hardness – It is due to the presence of soluble salts of magnesium and calcium in the form of chlorides and sulphates in water. It can be removed by washing soda or by ion exchange.
Ion exchange – Hardness of water can be removed by passing through an ion exchange bed like zeolites or polymer (resin) containing column.
Zeolites – Hydrated sodium alumino-silicates NaO.Al2O3.xSiO2.yH2O (x = 2-10, y = 2-6).
Premutit (or) zeolite process – The softening of water process by using complex structure represented as Na2 – Z with sodium as exchangeable cations.
Methods used to soften hard water – Chelating method and reverse osmosis.
Chelating method – EDTA is used in this.
Reverse osmosis – High pressure to force water through a semipermeable membrane.
Heavy water – D2O. It is present as one part in 5000 parts of ordinary water.
Preparation of D2O – It is prepared by continuous electrolysis of ordinary water. It undergoes exchange reactions.
Uses of D2O – D2O is used in isotopic effect, used as moderator in nuclear reactors, used to prepare deuterium compounds.
Hydrogen peroxide (H2O2) – It is a pale blue (almost colourless) liquid. It is prepared by the action of dilute acid with metal peroxide.
Uses of H2O2 – Oxidising agent, mild antiseptic, as a bleaching agent in textile, paper and hair-care industry and used as reducing agent.
Structure of H2O2 –
Hydrides – A binary hydride is a compound formed by hydrogen with other electropositive elements, e.g. LiH, MgH2.
Ternary hydrides – Compounds in which molecule is constituted by hydrogen and two types of elements, e.g. LiBH4, LiAlH4.
Types of hydrides –
| Ionic hydrides | Covalent hydride | Metallic hydride |
| Electropositive metal + hydrogen | Non-metals + Hydrogen | Metals + Hydrogen |
| Formation of hydride by transfer of electrons. | Formation of hydride by equal sharing of electrons. | Hydrogen occupies voids in metals. |
| e.g. LiH, CaH2 | e.g.CH4,SiH4 | e.g. ZnH2, TiH. |
Hydrogen sponge – Metal hydride as (PdH) Palladium hydride. It is formed by a chemical reaction but it behaves like a physical storage method, i.e., it is absorbed and released like a water sponge.
Hydrogen bonding – When a hydrogen atom is covalently bonded to a highly electronegative atom [ N (or) D (or) F], the bond is polarised in such a way that the hydrogen atom is able to form a weak bond between the hydrogen atom and the electronegative atom of a second molecule. This is called a hydrogen bond and denoted as dotted lines (…………….).
Types of hydrogen bonding – Intra molecular hydrogen bonding and inter molecular hydrogen bonding.
Intra molecular hydrogen bonding – These bonds are those which occur within one single molecule. e.g.T ortho-nitrophenol and salicylaldehyde.
Inter molecular hydrogen bonding – These bonds are formed between two separate molecules, e.g., Ammonia, water, hydrogen fluoride, acetic acid.
Gas hydrates – They are a kind of inclusion compounds, where gas molecules are trapped in a crystal lattice having voids of right size, with being chemically bonded, e.g., H3O+ in gas phase similar to methane hydrate.
Clatharate hydrate – Gas hydrates in which guest molecules are not bonded chemically but retained by the structure of host is called Clatharate hydrates, e.g., Methane hydrate CH4.20H2O.
Crystalline hydrates – In this type, hydrogen bonding is present. Water molecules serve to fill in the interstices and bind together structure, e.g., CuSO4.5H2O, Na2CO3.10H2O, FeSO4.7H2O.
The atomic bomb – The hydrogen bomb uses both nuclear fission and fusion is called atom bomb or Thermonuclear bombs.
The future fuel – Hydrogen is considered as a potential fuel as it is a clean burning fuel. This fuel is free from pollutants.