Students can Download Samacheer Kalvi 10th Science Model Question Paper 1 English Medium Pdf, Samacheer Kalvi 10th Science Model Question Papers helps you to revise the complete Tamilnadu State Board New Syllabus and score more marks in your examinations.
Tamil Nadu Samacheer Kalvi 10th Science Model Question Paper 1 English Medium
General Instructions:
- The question paper comprises of four parts
- You are to attempt all the questions in each part. An internal choice of questions is provided wherever applicable.
- All questions of Part I, II, III and IV are to be attempted separately.
- Question numbers 1 to 12 in Part I are Multiple Choice Questions of one mark each.
These are to be answered by writing the correct answer along with the corresponding option code. - Question numbers 13 to 22 in Part II are of two marks each. Any one question should be answered compulsorily.
- Question numbers 23 to 32 in Part III are of four marks each. Any one question should be answered compulsorily.
- Question numbers 33 to 35 in Part IV are of seven marks each. Draw diagrams wherever necessary.
Time: 3 Hours
Maximum Marks: 75
Part – I
(i) Answer all the questions. [12 × 1 = 12]
(ii) Choose the most suitable answer and write the code with the corresponding answer.
Question 1.
The unit of ‘g’ is ms-2. It can be also expressed as _______.
(a) cm s-1
(b) N kg-1
(c) N m2 kg-1
(d) cm2 s-2
Answer:
N kg-1
Question 2.
A convex lens forms a real, diminished point sized image at focus. Then the position of the object is at _____.
(a) focus
(b) infinity
(c) at 2f
(d) between f and 2 f
Answer:
(b) infinity
![]()
Question 3.
Velocity of sound is maximum in ________.
(a) Solid
(b) Liquid
(c) Gases
(d) All the above
Answer:
(a) Solid
Question 4.
The volume occupied by 4.4 g of CO2 at S.T.P _______.
(a) 22.4 litre
(b) 2.24 litre
(c) 0.24 litre
(d) 0.1 litre
Answer:
(b) 2.24 litre
Question 5.
Which of the following have inert gases 2 electrons in the outermost shell?
(a) He
(b) Ne
(c) Ar
(d) Kr
Answer:
(a) He
Question 6.
Deliquescence is due to ________.
(a) Strong affinity to water
(b) Less affinity of water
(c) Strong hatred to water
(d) Inertness to water
Answer:
(a) Strong affinity to water
![]()
Question 7.
Decrease in leucocytes is _______.
(a) Leucopenia
(b) Leukemia
(c) Anaemia
(d) None of the above
Answer:
(a) Leucopenia
Question 8.
Cancer cells are more easily damaged by radiations than normal cells because they are ______.
(a) Different in structure
(b) Non-dividing
(c) Mutated Cells
(d) Undergoing rapid division
Answer:
(d) Undergoing rapid division
Question 9.
The xylem and phloem arranged side by side on same radius is called ________.
(a) radial
(b) amphivasan
(c) conjoint
(d) amphicribral
Answer:
(c) conjoint
Question 10.
______ is the green coloured plastids.
(a) Chloroplast
(b) Chromoplast
(c) Leucoplast
(d) Stele
Answer:
(a) Chloroplast
![]()
Question 11.
The best way of direct dating fossils of recent origin is by ________.
(a) Radio-carbon method
(b) Uranium lead method
(c) Potassium-argon method
(d) Both (a) and (c)
Answer:
(a) Radio-carbon method
Question 12.
All files are stored in the _______.
(a) Box
(b) Pai
(c) Scanner
(d) Folder
Answer:
(d) Folder
Part – II
Answer any seven questions. (Q.No: 22 is compulsory) [7 × 2 = 14]
Question 13.
Define moment of a couple.
Answer:
When two equal and unlike parallel forces applied simultaneously at two distinct points constitute couple. A couple results in causes the rotation of the body. This rotating effect of a couple is known as moment of a couple.
![]()
Question 14.
Distinguish between the resistivity and conductivity of a conductor?
Answer:
| Resistivity | Conductivity |
| 1. It is the resistance of a conductor of unit length and unit area of cross section. | 1. It is the reciprocal of electrical resistivity of material. |
| 2. It’s unit is ohm metre | 2. Its unit is ohm–1 metre-1 or ohm metre-1 |
Question 15.
Why does sound travel faster on a rainy day than on a dry day?
Answer:
When humidity increases, the speed of sound increases. That is why you can hear sound from long distances clearly during rainy seasons.
Question 16.
What is rust? Give the equation for formation of rust?
Answer:
When iron is exposed to moist air, it forms a layer of brown hydrated ferric oxide on its surface. This compound is known as rust and the phenomenon of formation of rust is known as rusting.
![]()
Question 17.
Give an example each
(i) gas in liquid
(ii) solid in liquid
(iii) solid in solid
(iv) gas in gas.
Answer:
(i) Carbon-di-oxide dissolved in water (Soda water)
(ii) Sodium chloride dissolved in water.
(iii) Copper dissolved in gold (Alloy)
(iv) Mixture of Helium – Oxygen gases
![]()
Question 18.
How does leech respire?
Answer:
Respiration takes place through the skin in leech. The exchange of respiratory gases takes place by diffusion. Oxygen dissolved in water diffuses through the skin into haemocoelic fluid, while carbon dioxide diffuses out.
Question 19.
What is bolting? How can it be induced artificially?
Answer:
Treatment of rosette plants with gibberellin induces sudden shoot elongation followed by flowering is called bolting.
Question 20.
Match the following:

Answer:
(a) (iii)
(b) (i)
(c) (iv)
(d) (ii)
Question 21.
What is Atavism?
Answer:
The reappearance of ancestral characters in some individuals is called atavism. E.g. Presence of rudimentary tail in new born babies.
![]()
Question 22.
Calculate the velocity of a moving body of mass 7 kg whose linear momentum is 5 kg ms-1
Answer:
Given, p = 5 kg ms-1
m = 7 kg
p =m × v
v = \(\frac{p}{m}=\frac{5}{7}\)
∴ v = 0.71 ms-1
Part – III
Answer any seven questions (Q.No: 32 is compulsory) [7 × 4 = 28]
Question 23.
State and prove the law of conservation of linear momentum.
Answer:
(i) There is no change in the linear momentum of a system of bodies as long as no net external force acts on them.
(ii) Let us prove the law of conservation of linear momentum with the following illustration:
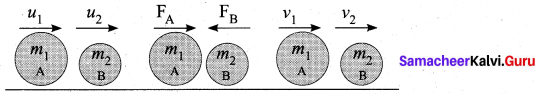
(iii) Let two bodies A and B having masses m1 and m2 move with initial velocity u1 and u2 in a straight line.
(iv) Let the velocity of the first body be higher than that of the second body. i.e., U1 > u2.
(v) During an interval of time t second, they tend to have a collision. After the impact, both of them move along the same straight line with a velocity v, and v2 respectively.
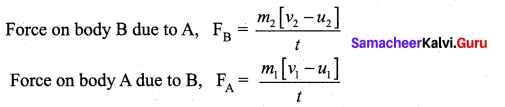
By Newton’s III law of motion
Action force = Reaction force
FA = -FB
\(\frac{m_{1}\left[v_{1}-u_{1}\right]}{t}=-\frac{m_{2}\left[v_{2}-u_{2}\right]}{t}\)
m1v1 – m1u1 = m2u2 + m2u2
m1v1 + m2v2 = m1u1 + m2u2
The above equation confirms in the absence of an external force, the algebraic sum of the momentum after collision is numerically equal to the algebraic sum of the momentum before collision. Hence the law of conservation linear momentum is proved.
![]()
Question 24.
(i) Explain about domestic electric circuits.
Answer:
- Electricity is distributed through the domestic electric circuits wired by the electricians.
- The first stage of the domestic circuit is to bring the power supply to the main-box from a distribution panel, such as a transformer.
- The important components of the main-box are: (i) a fuse box and (ii) a meter. The meter is used to record the consumption of electrical energy.
- The fuse box contains either a fuse wire or a miniature circuit breaker (MCB).
- The function of the fuse wire or a MCB is to protect the house hold electrical appliances from overloading due to excess current.
(ii) What connection is used in domestic appliances and why?
Answer:
- In domestic appliance is used parallel connection to avoid short circuit and breakage.
- One more advantage of the parallel connection of circuits is that each electric appliance gets an equal voltage.
Question 25.
(i) What is an echo?
Answer:
An echo is the sound reproduced due to the reflection of the original sound from various rigid surfaces such as walls, ceilings, surfaces of mountains, etc.
(ii) State two conditions necessary for hearing an echo.
Answer:
(i) The persistence of hearing for human ears is 0.1 second. This means that we can hear two sound waves clearly, if the time interval between the two sounds is atleast 0.1 s. Thus, the minimum time gap between the original sound and an echo must be 0.1 s.
(ii) The above criterion can be satisfied only when the distance between the source of sound and the reflecting surface would satisfy the following equation:

![]()
Question 26.
(i) What are the advantages of detergents over soaps?
Answer:
Detergents are better than soaps because they:
- can be used in both hard and soft water and can clean more effectively in hard water than soap.
- can also be used in saline and acidic water.
- do not leave any soap scum on the tub or clothes.
- dissolve freely even in cool water and rinse freely in hard water.
- can be used for washing woollen garments, where as soap cannot be used.
- have a linear hydrocarbon chain, which is biodegradable.
- are active emulsifiers of motor grease.
- do an effective and safe cleansing, keeping even synthetic fabrics brighter and whiter.
(b) Mention the disadvantages of detergents.
Answer:
- Some detergents having a branched hydrocarbon chain are not fully biodegradable by micro-organisms present in water. So, they cause water pollution.
- They are relatively more expensive than soap.
Question 27.
(a) Why does the reaction rate of a reaction increase on raising the temperature?
Answer:
On increasing temperature heat is supplied to the reactant. This energy breaks more bonds and thus speed up the chemical reaction. Foods kept at room temperature spoils – faster than that kept in the refrigerator.
(b) Explain the types of double displacement reactions with examples.
Answer:
There are two major classes of double displacement reactions. They are,
(i) Precipitation Reactions:
When aqueous solutions of two compounds are mixed, if they react to form an insoluble compound and a soluble compound, then it is called precipitation reaction.
Pb(NO3)2(aq) + 2KI(aq) → PbI2(s)↓+ 2KNO3(aq)
(ii) Neutralisation Reactions:
Another type of displacement reaction in which the acid reacts with the base to form a salt and water. It is called ‘neutralisation reaction’ as both acid and base neutralize each other.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(i)
![]()
Question 28.
How nerve impulses are transferred from one neuron to next neuron?
Answer:
All information from the environment are detected by the receptors, located in the sense organs such as the eyes, nose, skin, etc.
Information from the receptors is transmitted as electrical impulse and is received by the dendritic tips of the neuron. This impulse travels from the dendrite to the cell body and then along the axon to its terminal end.
On reaching the axonal end, it causes the nerve endings to release a chemical (neurotransmitter) which diffuses across a synapse and starts a similar electrical impulse in the dendrites of the next neuron, then to their cell body to be carried along the axon.
In this way, the electrical signal reaches the brain or spinal cord. The response from brain (or spinal cord) is similarly passed on to the effector organs such as the muscle or gland cell, that undergoes the desired response. The flow of nerve impulses from axon end of one neuron to dendrite of another neuron through a synapse is called synapatic transmission.
Question 29.
(a) How is dihybrid cross differ from monohybrid cross?
Monohybrid cross:
- It is a genetic cross, that involves a single pair of genes.
- In this cross, Parents differ by single trait (Eg : height)
- Monohybrid ratio in F2 generation is 3 : 1
Dihybrid cross:
- It is a genetic cross, that involves two pairs of genes, which are responsible for two traits.
- In this cross, the parents have two different independent traits. Eg: Flower colour, stem length
- The dihybrid ratio in F2 generation is 9 : 3 : 3 : 1
(b) What is cohesion?
Answer:
The force of attraction between molecules of water is called cohesion.
Question 30.
(a) Give an account on vascular bundle of dicot stem.
Answer:
The vascular bundles of dicot stem (Eg. sunflower) are conjoint, collateral, endarch and open. They are arranged in the form of a ring around the pith.
(b) Name the three basic tissue system in flowering plants.
Answer:
The three basic tissue system in flowering plants are
- Dermal or Epidermal tissue system
- Ground tissue system
- Vascular tissue system
Question 31.
(a) What are psychotropic drugs?
Answer:
The drugs which act on the brain and alter the behaviour, consciousness, power of thinking and perception, are called Psychotropic drugs. They are also called Mood altering drugs.
(b) State the applications of DNA fingerprinting technique.
Answer:
- DNA fingerprinting technique is widely used in forensic applications like crime investigation such as identifying the culprit.
- It is also used for paternity testing in case of disputes.
- It also helps in the study of genetic diversity of population, evolution and speciation.
![]()
Question 32.
(a) What are the applications of Doppler effect.
Answer:
(i) To measure the speed of an automobile:
- A source emitting electromagnetic wave is attached to a police car
- The wave is reflected by a moving source.
- From the frequency shift, the over speeding vehicles can be determined.
(ii) Tracking a satellite:
- The frequency of radio waves decreases as the satellite passes away from the Earth.
- By this change in frequency, the location of the satellites is determined.
(iii) RADAR:
- The radio waves are detected by the receivers.
- The frequency change tracks the speed and location of the aircraft’s.
(iv) SONAR:
- There is a frequency change between the sent signal and received signal.
- This helps to determine the speed of submariners and marine animals.
(ii) A solution is made from 25 mi of ethanol and 75 ml of water. Calculate the volume percentage.
Answer:
Volume of ethenol = 25 ml
Volume of water = 75 ml
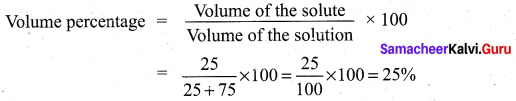
Part – IV
(1) Answer all the questions. [3 x 7 = 21]
(2) Each question carries seven marks.
(3) Draw diagram wherever necessary.
Question 33.
(a) (i) What are the characteristic features of heat energy transfer?
Answer:
- Heat always flows from a system of higher temperature to a system at lower temperature.
- The mass of a system is not altered when it is heated or cooled.
- Heat gained by the cold system is equal to heat lost by the hot system.
(ii) Explain linear expansion with diagram?
Answer:
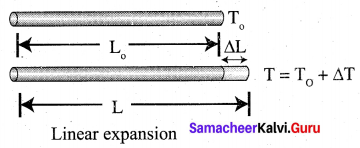
- When a body is heated or cooled, the length of the body changes due to change in its temperature.
- Then the expansion is said to be linear or longitudinal expansion.
- The ratio of increase in length of the body per degree rise in temperature to its unit length is called as the coefficient of linear expansion.
- It is different for different materials. Its SI unit is K-1.
- The equation is \(\frac{\Delta L}{L_{0}}\) = αLΔT
ΔL – Change in length (Final length – Original length)
L0 – Original length (Final temperature – Initial temperature)
αL – Coefficient of linear expansion
[OR]
(b) (i) In the circuit diagram given below, three resistors R1, R2 and R3 of 5 Ω, 10Ω and 20Ω respectively are connected as shown.
Calculate:
(A) Current through each resistor
(B) Total current in the circuit
(C) Total resistance in the circuit
Answer:
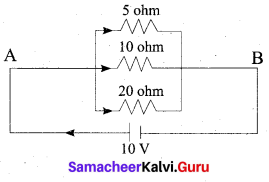
(A) Since the resistors are connected in parallel, the potential difference across each resistor
is same (i.e. V = 10V)
Therefore, the current through R1 is,
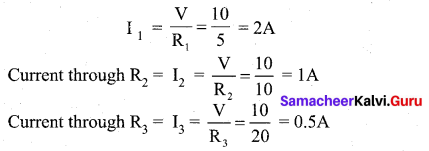
(B) Total current in the circuit,
I = I1 + I2 + I3
= 2 + 1 + 0.5 = 3.5 A
(C) Total resistance in the circuit
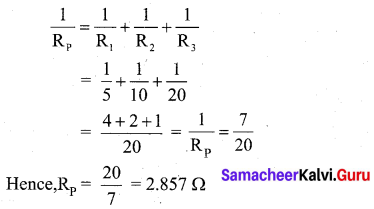
(ii) A charge of 12 coulomb flows through a bulb in 6 seconds. What is the current through the bulb?
Answer:
Charge Q = 12 C, Time t = 6 s. Therefore,
Current I = \(\frac{Q}{t}=\frac{12}{6}\) = 2A
![]()
Question 34.
(a) (i) Calculate the number of moles in 27 g of Al.
Answer:

(ii) Find the percentages of nitrogen in ammonia.
Answer:
Molar mass of NH3 = 1 × atomic mass of nitrogen + 3 × atomic mass of hydrogen
= (1 × 14) + (3 × 1)
= 14 + 3 = 17g

The percentage of nitrogen in ammonia = 82.35 %
(iii) Calculate the molecular mass of C2H6.
Answer:
Molecular mass of C2H6 = (2 × atomic mass of C) + (6 × atomic mass of H)
= (2 × 12) + (6 × 1)
= 24 + 6 = 30 g
[OR]
(b) Write notes on various factors affecting solubility.
Answer:
Factors affecting solubility : There are three main factors which govern the solubility of a solute. They are Nature of the solute and solvent, Temperature, and Pressure.
Nature of the solute and solvent:
- The nature of the solute and solvent plays an important role in solubility.
) Solubility is based on the phrase “like dissolves like.” The expression means that dissolving occurs when similarities exist between the solvent and the solute.
For example: Common salt is a polar compound and dissolves readily in polar solvent like water.
(iii) Non-polar compounds are soluble in non-polar solvents. For example, Sulphur dissolves in carbon di sulphide.
Effect of Temperature:
Solubility of Solids in Liquid:
- Generally, solubility of a solid solute in a liquid solvent increases with increase in temperature.
- In endothermic process, solubility increases with increase in temperature.
- In exothermic process, solubility decreases with increase in temperature.
Solubility of Gases in liquid:
Solubility of gases in liquid decrease with increase in temperature.
Effect of Pressure:
When the pressure is increased, the solubility of a gas in liquid increase. When pressure is increased, more gas molecules strike the surface of the liquid and mix into the solution.
![]()
Question 35.
(i) What is Cretinism?
Answer:
Cretinism: It is caused due to decreased secretion of thyroid hormone (thyroxine) in children. The conditions are stunted growth, mental defect, lack of skeletal development and deformed bones.
(ii) Write the characteristics of insect pollinated flowers?
Answer:
- Brightly coloured flowers, with smell and nectar.
- Pollen grain are larger in size, the exine is pitted, spiny so they can be adhered firmly on the sticky stigma.
(iii) What are the advantages of using bio-gas?
Answer:
The advantages of using biogas are as follows:
- It bums without smoke and therefore causing less pollution.
- An excellent way to get rid of organic wastes like bio-waste and sewage material.
- Left over slurry is a good manure rich in nitrogen and phosphorus.
- It is safe and convenient to use.
- It can reduce the amount of green house gases emitted.
[OR]
(b) (i) What is hermaphrodite?
Answer:
Hermaphrodite: If the male and female reproductive organs are present in the same individual, it is called hermaphrodite animal. Eg. Leech.
(ii) What are the effects of hybrid vigour?
Answer:
The superiority of the hybrid obtained by cross breeding is called as heterosis or hybrid vigour. Its effects in animal breeding are:
- Increased production of milk by cattle
- Increased production of egg by poultry
- High quality of meat is produced
- Increased growth rate in domesticated animals.