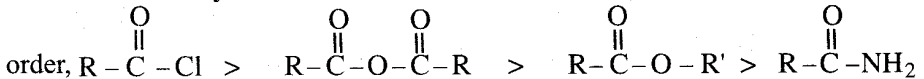Tamilnadu Samacheer Kalvi 11th Chemistry Notes Chapter 5 Alkali and Alkaline Earth Metals Notes
s-block elements – Group 1 and 2 elements are those in which the last electron enters the outermost s-orbital.
Group 1 elements – Alkali metals Li, Na, K, Rb, Cs and Fr with electronic configuration as noble gas [ns1].
Occurrence of Alkali metals
| Element | Occurrence |
| 1. Lithium | Spodume and lepidolite (Silicate minerals) |
| 2. Sodium | Rock salt (NaCl) |
| 3. Potassium | Nitre and camallite |
| 4. Rubidium and cesium | Minerals with other alkali metals |
| 5. Francium | Radioactive. It does not occur in nature |
Half-life of francium – 21 minutes.
Oxidation state of alkali metals – +1.
Atomic radii and ionic radii of alkali metals – On moving down the group increases and across the period decreases.
Periodic trends in alkali group – Reactivity, atomic radius, formation of electropositive ion and density increases down the group. Melting point and boiling point decreases down the group.
Distinctive behaviour of lithium – Extremely small size, greater polarizing power of ion, least electropositive character and non-availability of d-orbitals.
Chemical properties – Alkali metals are highly reactive and reacts with oxygen, hydrogen, halogen and liquid ammonia.
Uses of alkali metals – Oxidizing agents (oxide of alkali metals), strong bases (hydroxides of alkali metals), sodium and potassium ions perform important biological functions such as ion balance and nerve impulse conduction.
Fruits rich in potassium – Avocadoes, potatoes and bananas.
Group 2 elements – Alkaline earth elements with general electronic configuration as [noble gases] ns2. They are Be, Mg, Ca, Sr, Ba and Ra.
Alkaline earth metals – Except Be, all other oxides and hydroxides are alkaline in nature.
Occurrence of alkaline earth metals –
| Element | Occurrence |
| 1. Beryllium | Rare |
| 2. Radium | The rarest |
| 3. Magnesium and calcium | Rocks and minerals |
| 4. Magnesium | 8th most abundant element and occur as camallite, magnesite and dolomite |
| 5. Calcium | 5th most abundant element and occur as chalk, limestone and gypsum |
| 6. Strontium | Celestite and strontianite |
| 7. Barium | Less common and occur as barite |
Pyrotechnics – Alkaline earth metals are used to produce colours in firework shows.
Oxidation state of alkaline earth metals – +2.
Trends in periodic properties –
- Atomic and ionic radii increases down the group.
- Ionization enthalpy and hydration enthalpy decreases down the group.
Anomalous behavior of beryllium – (i) Small size (ii) high polarizing power (iii) high electronegativity (iv) absence of vacant d-orbital (v) high ionization enthalpy.
Chemical properties – Alkali metals react with H., and halogens.
Uses of beryllium –
- Used as radiation windows for X-ray tube and X-ray detectors.
- As sample holder in X-ray emission studies.
- Used to build the beam pipe in accelerators.
- Used in detectors.
Uses of magnesium –
- For the removal of sulfur from iron and steel.
- For the refining of titanium.
- Used as photoengrave plates in printing industry.
- Magnesium alloys are used in airplanes and missile construction.
- Mg ribbon is used in Grignard reagent synthesis.
- As desiccant.
- As sacrificial anode in controlling galvanic corrosion.
- As a reducing agent.
- Mg + A1 alloy used in fabrication and welding.
Uses of calcium –
- Used as a reducing agent in the metallurgy of uranium, zirconium and thorium.
- As a deoxidizer, desulfurizer or decarbonizer for ferrous and non-ferrous alloys.
- As a getter in vacuum tubes.
- In making of cements and mortars.
- In dehydrating oils.
- In fertilizers, concrete and making Plaster of Paris.
Uses of strontium –
- 90Sr is used in cancer therapy.
- 87Sr / 86Sr ratio is used in marine investigations as well as in teeth, tracking animal migrations or in criminal forensics.
- Used in dating of rocks.
- Used as a radioactive tracer.
Uses of barium –
- Used in metallurgy, pyrotechnics, petroleum mining and radiology.
- Deoxidizer in copper refining.
- Ba + Ni alloy is used in electron tubes and in spark plug electrodes.
- As a scavenger to remove oxygen and other gases in television and electronic tubes.
- 133Ba is used as a source in the calibration of gamma ray detector.
Uses of radium – Used in self-luminous paints for watches, nuclear panels, aircraft switches, clocks and instrument dials.
Chemical properties of alkaline earth metals –
- They form monoxides and peroxide with oxygen.
- The oxides of alkaline earth metals react with water to give hydroxides.
- They form halides when react with halogens.
- Alkaline earth metals form salts of oxo-acids such as carbonates, sulphates and nitrates.
Important compounds of calcium –
Quick lime – CaO
Slaking of lime – The process of addition of limited amount of water breaks the lump of lime is called slaking of lime.
Uses of quick lime –
- To manufacture cement, mortar and glass.
- To manufacture sodium carbonate and slaked lime.
- In the purification of sugar.
- As drying agent.
Slaked lime – Ca(OH)2
Bleaching powder – Ca(OCl)2
Uses of slaked lime –
- In the preparation of mortar, a building material.
- In white wash.
- In glass making and in tanning industry.
- For the preparation of bleaching powder and in purification of sugar.
Gypsum – CaSO4.2H2O
Desert Rose – Gypsum crystals are found to occur in a form that resembles the petals of a flower and this occur in desert terrains.
Alabaster – A variety of gypsum and valued as an ornamental stone.
Plaster of Paris – CaSO4.1/2 H2O
Uses of gypsum –
- It has been used by the sculptors.
- To prepare Plaster of Paris.
- Used in making dry walls or plaster boards.
- It is used in making surgical and orthopedic casts, casting molds and surgical splints.
- It is used in agriculture as a soil additive, conditioner and fertilizer.
- It is used in toothpaste, shampoo and hair products.
Calcium sulfate – Acts as a coagulator in making tofu. It is used in baking, as a dough conditioner. It is used to treat upset stomach and eczema.
Gypsum – It is used as a hardening retarder to control the speed at which concrete sets.
Satin spar – It is a variety of gypsum, used as an ornamental stone, while alabaster is used for sculpting.
Gypsum – It is used to give colour to cosmetics and drugs. It can be found in canned vegetables, flour, ice cream, blue cheese and white bread. It is mainly used in wine making.
Uses of Plaster of Paris –
- It is used in building industry as well as plasters.
- It is used for immobilizing the affected part of organ, where there is a bone fracture or sprain.
- It is also used in dentistry, making casts of statues and busts.
Biological importance of magnesium and calcium –
- An adult body contains about 25 g of Mg and 1200 g of Ca. The daily requirement in the human body has been estimated to be 200 – 300 mg.
- All enzymes transfer requires magnesium as the co-factor. The main pigment of chlorophyll is magnesium.
- 99% of body calcium is present in bones and teeth. It also play important roles in neuromuscular function, intemeuronal transmission, cell membrane integrity and blood coagulation.
- The calcium concentration is maintained by two hormones – calcitonin and parathyroid hormones.
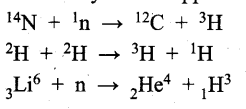
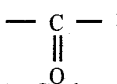 is called carbonyl compounds. They plays an important role in the metabolic process. Carbonyl compounds are important constituents of fabrics, plastics and drugs.
is called carbonyl compounds. They plays an important role in the metabolic process. Carbonyl compounds are important constituents of fabrics, plastics and drugs.